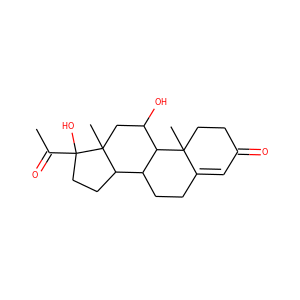
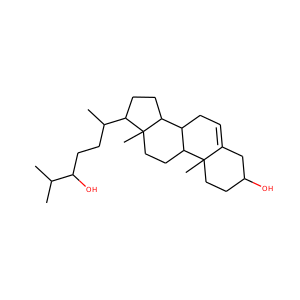
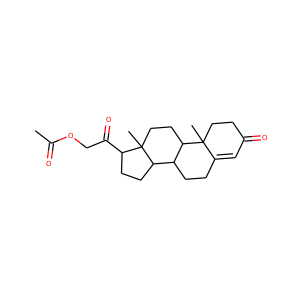
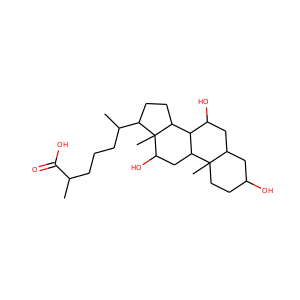
| Weight | 318.501 g/mol |
|---|---|
| Formula | C21H34O2 |
| Hydrogen Acceptors | 2 |
| Hydrogen Donors | 1 |
| Aromatic Rings | 0 |
| Rotatable Bonds | 1 |
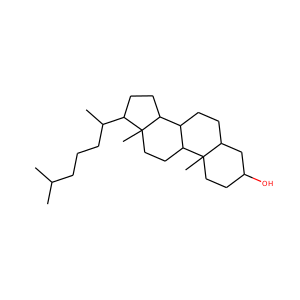
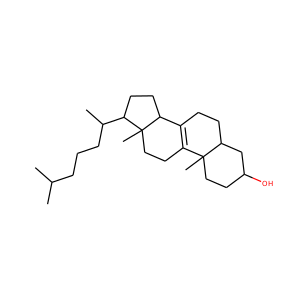
3-Hydroxypregnan-20-one (128-20-1, 4406-35-3)
"What do you need help with?"

 LabBot
LabBot
- Sigma-Aldrich - Search for 3-Hydroxypregnan-20-one
- Fisher Scientific - Search for 3-Hydroxypregnan-20-one
- TCI - Search for 3-Hydroxypregnan-20-one
- Neurosteroid Analogues. 6. The Synthesis and GABAA Receptor Pharmacology of Enantiomers of Dehydroepiandrosterone Sulfate, Pregnenolone Sulfate, and (3,5)-3-Hydroxypregnan-20-one Sulfate (Journal of Medicinal Chemistry, 1998)
- Neurosteroid analogues. 9. Conformationally constrained pregnanes: Structure-activity studies of 13,24-cyclo-18,21-dinorcholane analogues of the GABA modulatory and anesthetic steroids (3, 5)- and (3, 5)-3-hydroxypregnan-20-one (Journal of Medicinal Chemistry, 2003)
- Neurosteroid Analogues. 8. StructureActivity Studies of N-Acylated 17a-Aza-D-homosteroid Analogues of the Anesthetic Steroids (3,5)- and (3,5)-3-Hydroxypregnan-20-one (Journal of Medicinal Chemistry, 2000)
- Neurosteroid Analogues. 10. The Effect of Methyl Group Substitution at the C-6 and C-7 Positions on the GABA Modulatory and Anesthetic Actions of (3,5)- and (3,5)-3-Hydroxypregnan-20-one (Journal of Medicinal Chemistry, 2005)
- Ethanol alters local cellular levels of (3,5)-3-hydroxypregnan-20-one (3,5-THP) independent of the adrenals in subcortical brain regions. (Neuropsychopharmacology, 2014)
- Voluntary ethanol consumption reduces GABAergic neuroactive steroid (3,5)3-hydroxypregnan-20-one (3,5-THP) in the amygdala of the cynomolgus monkey. (Addiction Biology, 2017)
-
Predict GHS Hazards for Any Chemical in silico.
Including Acute Oral Tox, Skin Sensitization, Eye Irritation, Aquatic Tox, & more. -
SMILESCC(=O)C1CCC2C1(CCC3C2CCC4C3(CCC(C4)O)C)C
-
InChIKeyAURFZBICLPNKBZ-UHFFFAOYSA-N
- Pubchem - 3-Hydroxypregnan-20-one
- Wikipedia - Allopregnanolone
Allopregnanolone, also known as 5-pregnan-3-ol-20-one or 3,5-tetrahydroprogesterone (3,5-THP), as well as brexanolone (USAN), is an endogenous inhibitory pregnane neurosteroid. It is synthesized from progesterone, and is a potent positive allosteric modulator of the action of -amininobutyric acid (GABA) at GABAA receptor. Allopregnanolone has effects similar to those of other positive allosteric modulators of the GABA action at GABAA receptor such as the benzodiazepines, including anxiolytic, sedative, and anticonvulsant activity.
Suppliers
Most-cited Publications
Safety
Alternate Names
External Links
Similar Compounds
17-Hydroxypregnenolone
(387-79-1)
1 alternate name
3 suppliers
LITHOCHOLIC ACID
(434-13-9)
1 alternate name
3 suppliers
Isolithocholic acid
(1534-35-6)
1 alternate name
3 suppliers
Dihydrocholesterol
(80-97-7)
1 alternate name
3 suppliers
Lathosterol
(80-99-9)
1 alternate name
3 suppliers
DESMOSTEROL
(313-04-2)
1 alternate name
3 suppliers
Zymosterol
(128-33-6)
1 alternate name
3 suppliers
Zymostenol
(566-97-2)
1 alternate name
3 suppliers
27-hydroxycholesterol
(20380-11-4)
1 alternate name
3 suppliers
estradiol valerate
(979-32-8)
1 alternate name
3 suppliers
7-Dehydrocholesterol
(434-16-2)
1 alternate name
3 suppliers
Norethindrone acetate
(38673-38-0, 51-98-9)
1 alternate name
3 suppliers
19-Norethindrone acetate
(51-98-9)
1 alternate name
3 suppliers
hydroxyprogesterone
(604-09-1, 68-96-2)
1 alternate name
3 suppliers
17alpha-Hydroxypregn-4-ene-3,20-dione
(604-09-1)
1 alternate name
3 suppliers
21-Deoxyhydrocortisone
(641-77-0)
1 alternate name
3 suppliers
Cerebrosterol
(474-73-7)
1 alternate name
3 suppliers
22R-hydroxycholesterol
(22348-64-7, 17954-98-2)
1 alternate name
3 suppliers
STIGMASTEROL
(83-48-7)
1 alternate name
3 suppliers
BETA-SITOSTEROL
(68555-08-8, 19044-06-5, 83-46-5)
1 alternate name
3 suppliers
20-beta-Progerol
(145-15-3, 145-14-2)
1 alternate name
3 suppliers
Glycolithocholic acid
(474-74-8)
1 alternate name
3 suppliers
3,7-Dihydroxy-12-oxocholanoic acid
(2458-08-4)
1 alternate name
3 suppliers
7-Ketodeoxycholic acid
(911-40-0)
1 alternate name
3 suppliers
4,4-Dimechol-8,14,24-trienol
(64284-64-6)
1 alternate name
3 suppliers
Melengestro acetate
(425-51-4, 595-33-5)
1 alternate name
3 suppliers
Tetrahydrodeoxycorticosterone
(567-03-3, 567-02-2)
1 alternate name
3 suppliers
53-00-9
(53-00-9)
1 alternate name
3 suppliers
testosterone enanthate
(315-37-7)
1 alternate name
3 suppliers
Methyl 12-hydroxyoctadecanoate
(141-23-1)
1 alternate name
3 suppliers
12-Hydroxystearic acid
(106-14-9, 36377-33-0, 18417-00-0, 27924-99-8, 5762-36-7)
1 alternate name
3 suppliers
ISOPHYTOL
(505-32-8)
1 alternate name
2 safety hazards
3 suppliers
BUTYL STEARATE
(123-95-5, 68154-28-9)
1 alternate name
3 suppliers
Methyl ricinoleate
(141-24-2)
1 alternate name
3 suppliers
methyl 12-hydroxyoctadec-9-enoate
(141-24-2)
1 alternate name
3 suppliers
NEROLIDOL
(40716-66-3, 7212-44-4, 142-50-7)
1 alternate name
3 suppliers
PHYTANIC ACID
(14721-66-5)
1 alternate name
3 suppliers
phytosphingosine
(554-62-1)
1 alternate name
3 suppliers
Deoxycorticosterone acetate
(56-47-3)
1 alternate name
3 suppliers
Phytol
(150-86-7, 102608-53-7)
1 alternate name
3 suppliers
NEROLIDOL
(1119-38-6, 7212-44-4, 142-50-7)
1 alternate name
3 suppliers
pristanic acid
(1189-37-3)
1 alternate name
3 suppliers
ETHYL STEARATE
(111-61-5)
1 alternate name
3 suppliers
cis-Nerolidol
(3790-78-1, 7212-44-4, 142-50-7)
1 alternate name
3 suppliers
Coprocholic acid
(547-98-8)
1 alternate name
3 suppliers
CAS Directory