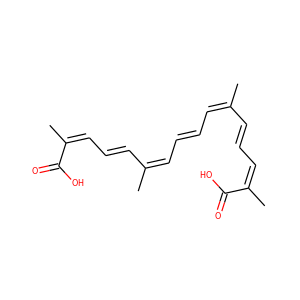
| Weight | 328.408 g/mol |
|---|---|
| Formula | C20H24O4 |
| Hydrogen Acceptors | 2 |
| Hydrogen Donors | 2 |
| Aromatic Rings | 0 |
| Rotatable Bonds | 8 |
Crocetin (27876-94-4)
crocetin sodium salt
 Score:
#2753 in Biochemistry
,
#5194 in Biology
,
#6802 in Chemistry
Score:
#2753 in Biochemistry
,
#5194 in Biology
,
#6802 in Chemistry
"What do you need help with?"

 LabBot
LabBot
- Sigma-Aldrich - Search for Crocetin
- Fisher Scientific - Search for Crocetin
- TCI - Search for Crocetin
- ANTI-INFLAMMATORY EFFECTS OF CROCIN AND CROCETIN IN RAT BRAIN MICROGLIAL CELLS (European Journal of Pharmacology, 2010)
- Antihyperlipidemic effect of crocin isolated from the fructus of Gardenia jasminoides and its metabolite Crocetin. (Biological & Pharmaceutical Bulletin, 2005)
- Neuroprotection by crocetin in a hemi-parkinsonian rat model (Pharmacology, Biochemistry and Behavior, 2005)
- Crocetin protects against oxidative damage in rat primary hepatocytes. (Cancer Letters, 1995)
- CROCETIN, DIMETHYLCROCETIN, AND SAFRANAL BIND HUMAN SERUM ALBUMIN: STABILITY AND ANTIOXIDATIVE PROPERTIES (Journal of Agricultural and Food Chemistry, 2007)
- Crocetin from Saffron: An Active Component of an Ancient Spice (Critical Reviews in Food Science and Nutrition, 2004)
-
Predict GHS Hazards for Any Chemical in silico.
Including Acute Oral Tox, Skin Sensitization, Eye Irritation, Aquatic Tox, & more. - crocetin sodium salt
-
SMILESCC(=CC=CC=C(C)C=CC=C(C)C(=O)O)C=CC=C(C)C(=O)O
-
InChIKeyPANKHBYNKQNAHN-UHFFFAOYSA-N
- Pubchem - Crocetin
- Wikipedia - transcrocetinate
Crocetin is a natural apocarotenoid dicarboxylic acid that is found in the crocus flower and Gardenia jasminoides (fruits). It forms brick red crystals with a melting point of 285C. The chemical structure of crocetin forms the central core of crocin, the compound responsible for the color of saffron.
Suppliers
Most-cited Publications
Areas of Application
Safety
Alternate Names
External Links
Similar Compounds
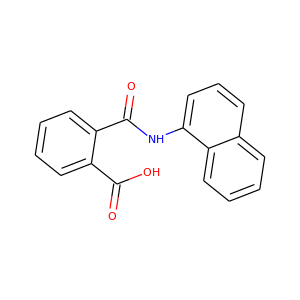
NAPTALAM
(132-66-1)
2 alternate names
3 suppliers
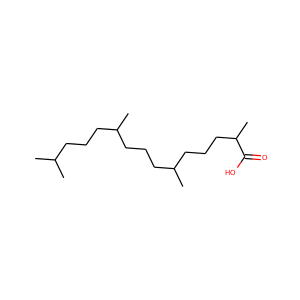
pristanic acid
(1189-37-3)
2 alternate names
3 suppliers
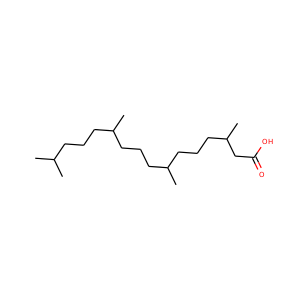
PHYTANIC ACID
(14721-66-5)
2 alternate names
3 suppliers
27955-94-8
(27955-94-8)
2 alternate names
3 safety hazards
3 suppliers
Amineptine
(57574-09-1)
2 alternate names
3 suppliers
91-48-5
(91-48-5, 3368-16-9)
2 alternate names
3 suppliers
Octadecanedioic acid
(871-70-5)
2 alternate names
3 suppliers
Chlorthal
(2136-79-0)
2 alternate names
3 suppliers
Diisooctyl phthalate
(27554-26-3)
2 alternate names
3 suppliers
ISOPHYTOL
(505-32-8)
2 alternate names
2 safety hazards
3 suppliers
3-Hydroxyflavone
(577-85-5)
2 alternate names
3 suppliers
Bisphenol B
(77-40-7)
2 alternate names
3 suppliers
Peramivir
(1041434-82-5, 229614-55-5, 229614-56-6, 330600-85-6)
2 alternate names
3 suppliers
Dinopol ido
(1330-96-7)
2 alternate names
3 suppliers
2-(Benzoylamino)benzoic acid
(579-93-1)
2 alternate names
3 suppliers
2,2'-Dithiodibenzoic acid
(119-80-2)
2 alternate names
3 suppliers
LINALYL ANTHRANILATE
(7149-26-0)
2 alternate names
3 suppliers
DIISONONYL PHTHALATE
(28553-12-0, 68515-48-0)
2 alternate names
3 suppliers
ERUCIC ACID
(1072-39-5, 112-86-7)
2 alternate names
3 suppliers
norethynodrel
(68-23-5)
2 alternate names
3 suppliers
RACTOPAMINE
(97825-25-7)
2 alternate names
3 suppliers
METALAXYL
(57837-19-1)
2 alternate names
2 safety hazards
3 suppliers
Octabenzone
(1843-05-6)
2 alternate names
3 suppliers
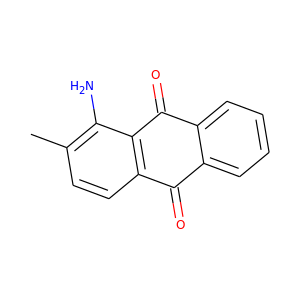
1-AMINO-2-METHYLANTHRAQUINONE
(82-28-0)
2 alternate names
3 suppliers
Phenmedipham
(13684-63-4)
2 alternate names
3 safety hazards
3 suppliers
Pararosaniline
(479-73-2)
2 alternate names
3 suppliers
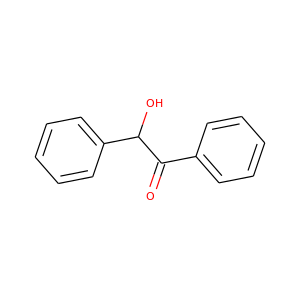
BENZOIN
(119-53-9, 8050-35-9, 9000-72-0, 9000-05-9, 579-44-2)
2 alternate names
3 suppliers
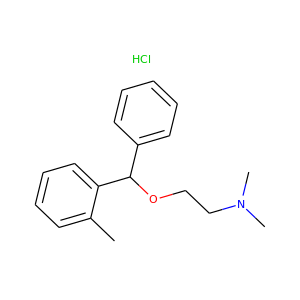
Orphenadrine hydrochloride
(341-69-5)
2 alternate names
3 suppliers
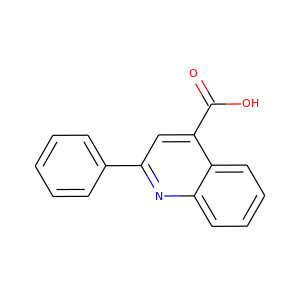
Cinchophen
(132-60-5)
2 alternate names
3 suppliers
DIISOOCTYL ADIPATE
(105-96-4, 1330-86-5)
2 alternate names
3 suppliers
CP 47497
(70434-82-1)
2 alternate names
3 suppliers
Arlacel 60
(76169-00-1, 1338-41-6)
2 alternate names
3 suppliers
4376-20-9
(4376-20-9)
2 alternate names
3 suppliers
SORBITAN MONOSTEARATE
(76169-00-1, 1338-41-6)
2 alternate names
3 suppliers
CAS Directory