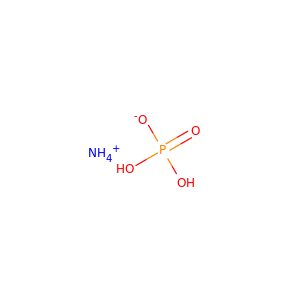
| Weight | 132.056 g/mol |
|---|---|
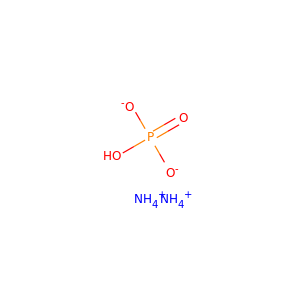
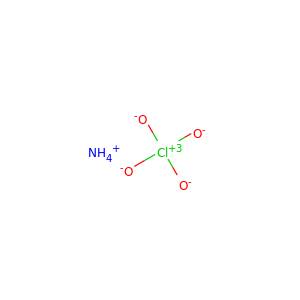
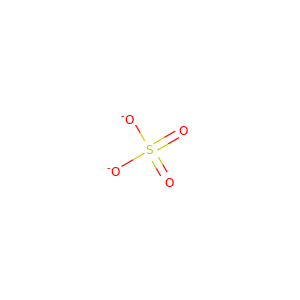
| Formula | H9N2O4P |
| Hydrogen Acceptors | 3 |
| Hydrogen Donors | 3 |
| Aromatic Rings | 0 |
| Rotatable Bonds | 0 |
DIAMMONIUM PHOSPHATE (7783-28-0)
ammonium phosphate · monoammonium phosphate · diammonium hydrogen phosphate
 Score:
#910 in Physics
,
#2096 in Biology
,
#2574 in Chemistry
Score:
#910 in Physics
,
#2096 in Biology
,
#2574 in Chemistry
"What do you need help with?"

 LabBot
LabBot
- Sigma-Aldrich - Search for DIAMMONIUM PHOSPHATE
- Fisher Scientific - Search for DIAMMONIUM PHOSPHATE
- TCI - Search for DIAMMONIUM PHOSPHATE
- COMPARISON OF THE EFFECTS OF A LOCALISED SUPPLY OF PHOSPHATE, NITRATE, AMMONIUM AND POTASSIUM ON THE GROWTH OF THE SEMINAL ROOT SYSTEM, AND THE SHOOT, IN BARLEY (New Phytologist, 1975)
- Use of Trialkyl Ammonium Phosphate (TAAP) Buffers in Reverse Phase HPLC for High Resolution and High Recovery of Peptides and Proteins (Journal of Liquid Chromatography & Related Technologies, 1978)
- Conditions influencing the precipitation of magnesium ammonium phosphate. (Water Research, 2001)
- Effect of biochar amendment on sorption and leaching of nitrate, ammonium, and phosphate in a sandy soil. (Chemosphere, 2012)
- Use of diammonium phosphate to reduce heavy metal solubility and transport in smelter-contaminated soil. (Journal of Environmental Quality, 2001)
- The diffusion coefficients of sulfate, ammonium, and phosphate ions in anoxic marine sediments (Limnology and Oceanography, 1980)
-
Predict GHS Hazards for Any Chemical in silico.
Including Acute Oral Tox, Skin Sensitization, Eye Irritation, Aquatic Tox, & more. - ammonium phosphate
- monoammonium phosphate
- diammonium hydrogen phosphate
- ammonium hydrogen phosphate
- ammonium orthophosphate
- triammonium phosphate
- monobasic ammonium phosphate
- ammonium phosphate ((NH4)3PO4)
- primary ammonum phosphate
- ammonium phosphate, dibasic
- tribasic ammonium phosphate
-
SMILES[NH4+].[NH4+].OP(=O)([O-])[O-]
-
InChIKeyMNNHAPBLZZVQHP-UHFFFAOYSA-N
- Pubchem - DIAMMONIUM PHOSPHATE
- Wikipedia - dibasicammonium phosphate
Diammonium phosphate (DAP) (chemical formula (NH4)2HPO4, IUPAC name diammonium hydrogen phosphate) is one of a series of water-soluble ammonium phosphate salts that can be produced when ammonia reacts with phosphoric acid. Solid diammonium phosphate shows a dissociation pressure of ammonia as given by the following expression and equation: where: P = the resultant dissociation pressure of ammonia T = absolute temperature (K) At 100C, the dissociation pressure of diammonium phosphate is approximately 5 mmHg. Accordingly, to MSDS of DiammoniumPhosphate from CF Industries inc.
Suppliers
Most-cited Publications
Areas of Application
Safety
Trends
Alternate Names
External Links
Similar Compounds
Ammonium acid arsenate
(7784-44-3)
12 alternate names
3 suppliers
AMMONIUM NITRATE
(54378-28-8, 6484-52-2)
12 alternate names
3 safety hazards
3 suppliers
AMMONIUM SULFATE
(7704-34-9, 7783-20-2, 68187-17-7, 7783-20-2, 68081-96-9)
12 alternate names
3 suppliers
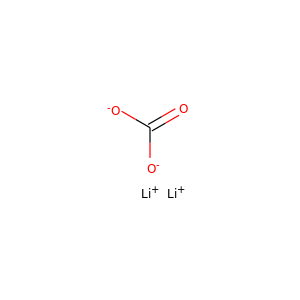
LITHIUM CARBONATE
(554-13-2, 7439-93-2, 10377-37-4, 554-13-2)
12 alternate names
4 safety hazards
3 suppliers
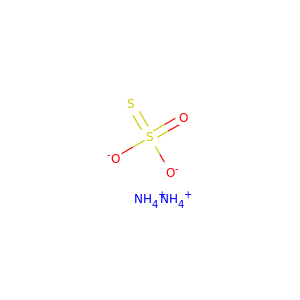
AMMONIUM THIOSULFATE
(7783-18-8)
12 alternate names
3 suppliers
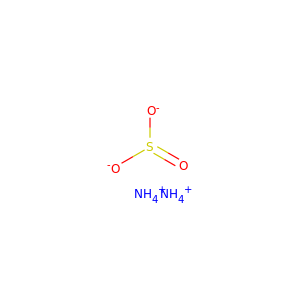
AMMONIUM SULFITE
(10196-04-0)
12 alternate names
3 suppliers
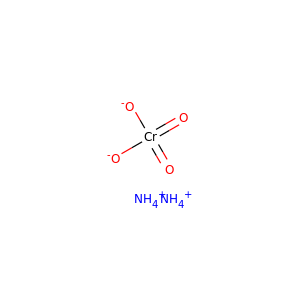
AMMONIUM CHROMATE
(52110-72-2, 7788-98-9)
12 alternate names
3 suppliers
BERYLLIUM PHOSPHATE
(13598-15-7)
12 alternate names
3 suppliers
Ammonium dihydrogen phosphate
(7722-76-1)
12 alternate names
3 suppliers
AMMONIUM SULFAMATE
(7773-06-0, 13765-36-1)
12 alternate names
2 safety hazards
3 suppliers
Hydroxylamine sulfate (2:1)
(10039-54-0)
12 alternate names
5 safety hazards
3 suppliers
nitrate
(14797-55-8)
12 alternate names
3 suppliers
AMMONIUM PERCHLORATE
(7790-98-9)
12 alternate names
3 safety hazards
3 suppliers
Dinitrogen tetroxide
(10544-72-6, 10102-44-0)
12 alternate names
5 safety hazards
3 suppliers
phosphate
(14265-44-2)
12 alternate names
3 suppliers
diphosphate
(2466-09-3, 14000-31-8)
12 alternate names
3 suppliers
Beryllium sulfate
(13510-49-1)
12 alternate names
3 suppliers
AMMONIUM BISULFITE
(10192-30-0)
12 alternate names
1 safety hazard
3 suppliers
Lithium peroxide (Li2(O2))
(12031-80-0)
12 alternate names
3 suppliers
NITRIC ACID
(68412-17-9, 7697-37-2, 7697-37-3, 12507-77-6, 7697-37-2)
12 alternate names
4 safety hazards
3 suppliers
selenate
(14124-68-6)
12 alternate names
3 suppliers
Ammonium metavanadate
(7803-55-6)
12 alternate names
4 safety hazards
3 suppliers
Ammonium persulfate
(7727-54-0)
12 alternate names
7 safety hazards
3 suppliers
LITHIUM CHROMATE
(14307-35-8)
12 alternate names
5 safety hazards
3 suppliers
Guanidine nitrate
(52470-25-4, 506-93-4)
12 alternate names
2 safety hazards
3 suppliers
1-Methyl-3-nitro-1-nitrosoguanidine
(70-25-7)
12 alternate names
6 safety hazards
3 suppliers
Nitroguanidine
(556-88-7)
12 alternate names
1 safety hazard
3 suppliers
sodium sulfide
(1313-82-2)
12 alternate names
4 safety hazards
3 suppliers
Ammonium hexafluorosilicate
(16919-19-0)
12 alternate names
4 safety hazards
3 suppliers
UREA NITRATE
(124-47-0)
12 alternate names
2 safety hazards
3 suppliers
1-CHLORO-1-NITROPROPANE
(600-25-9)
12 alternate names
3 safety hazards
3 suppliers
2-CHLORO-2-NITROPROPANE
(594-71-8)
12 alternate names
3 suppliers
trisodium phosphorus(3-)
(12058-85-4)
12 alternate names
3 suppliers
sulfate
(14808-79-8, 52037-76-0, 18785-72-3)
12 alternate names
3 suppliers
Dinitrogen trioxide
(10544-73-7)
12 alternate names
3 suppliers
2-nitropropane
(79-46-9)
12 alternate names
3 safety hazards
3 suppliers
SILVER NITRATE
(7761-88-8)
12 alternate names
5 safety hazards
3 suppliers
SODIUM NITRATE
(7631-99-4)
12 alternate names
3 safety hazards
3 suppliers
LEAD CHROMATE
(7758-97-6, 11119-70-3, 15804-54-3)
12 alternate names
7 safety hazards
3 suppliers
NITROMETHANE
(75-52-5)
12 alternate names
5 safety hazards
3 suppliers
ZINC CHROMATE
(13530-65-9)
12 alternate names
7 safety hazards
3 suppliers
NITROETHANE
(79-24-3)
12 alternate names
5 safety hazards
3 suppliers
bronopol
(52-51-7)
12 alternate names
7 safety hazards
3 suppliers
CALCIUM CHROMATE
(13765-19-0, 12205-18-4, 53568-70-0)
12 alternate names
9 safety hazards
3 suppliers
1-NITROPROPANE
(108-03-2)
12 alternate names
4 safety hazards
3 suppliers
sodium azide
(26628-22-8)
12 alternate names
5 safety hazards
3 suppliers
STRONTIUM CHROMATE
(7789-06-2)
12 alternate names
8 safety hazards
3 suppliers
Propyl nitrate
(627-13-4)
12 alternate names
2 safety hazards
3 suppliers
3-nitropropionic acid
(504-88-1)
12 alternate names
3 suppliers
BARIUM CHROMATE
(10294-40-3)
12 alternate names
5 safety hazards
3 suppliers
Hydrazine sulfate
(1184-66-3, 10034-93-2)
12 alternate names
6 safety hazards
3 suppliers
Lead tungstate
(7759-01-5)
12 alternate names
3 suppliers
Thallium nitrate
(10102-45-1, 16901-76-1)
12 alternate names
4 safety hazards
3 suppliers
Dimethylarsinate
(15132-04-4)
12 alternate names
3 suppliers
ETHYL NITRATE
(625-58-1)
12 alternate names
1 safety hazard
3 suppliers
Chromium chromate (H2CrO4)
(41261-95-4)
12 alternate names
3 suppliers
MERCUROUS NITRATE
(10415-75-5)
12 alternate names
5 safety hazards
3 suppliers
TRIMETHYLAMINE N-OXIDE
(1184-78-7)
12 alternate names
3 suppliers
Lead diazide
(13424-46-9)
12 alternate names
7 safety hazards
3 suppliers
Calcium metasilicate
(13983-17-0, 1344-95-2, 99328-53-7, 10101-39-0)
12 alternate names
3 suppliers
METHYLAZOXYMETHANOL
(590-96-5)
12 alternate names
3 suppliers
Potassium phosphide (K3P)
(20770-41-6)
12 alternate names
3 suppliers
4-Nitropyridine N-oxide
(1124-33-0)
12 alternate names
3 suppliers
SODIUM DIACETATE
(126-96-5)
12 alternate names
3 suppliers
2-Ethylhexyl sulfate
(126-92-1)
12 alternate names
3 suppliers
BERYLLIUM SULFATE TETRAHYDRATE
(7787-56-6 (tetrahydrate), 7787-56-6)
12 alternate names
5 safety hazards
3 suppliers
Hydrogen sulfate
(7664-93-9, 14996-02-2)
12 alternate names
3 suppliers
Permanganate
(14333-13-2)
12 alternate names
3 suppliers
ISOPROPYL NITRATE
(1712-64-7)
12 alternate names
3 suppliers
1-NITROHEXANE
(646-14-0, 25495-95-8)
12 alternate names
3 suppliers
thiosulfate
(14383-50-7)
12 alternate names
3 suppliers
1-NITROBUTANE
(627-05-4)
12 alternate names
3 suppliers
Amyl nitrate
(1002-16-0)
12 alternate names
3 suppliers
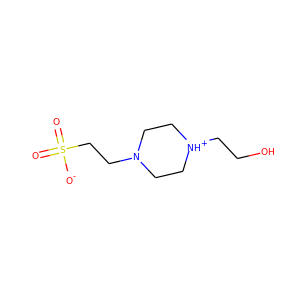
1-Piperazineethanesulfonic acid, 4-(2-hydroxyethyl)-
(7365-45-9)
12 alternate names
3 suppliers
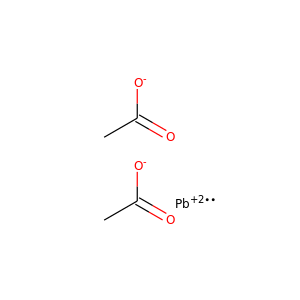
Lead diacetate
(15347-57-6, 301-04-2, 301-04-2, 6080-56-4)
12 alternate names
5 safety hazards
3 suppliers
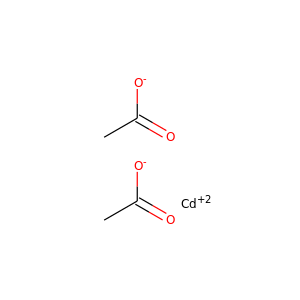
cadmium acetate
(5743-04-4, 543-90-8)
12 alternate names
8 safety hazards
3 suppliers
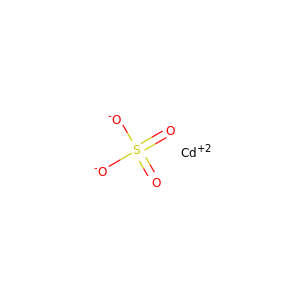
Cadmium sulfate
(13477-21-9, 10124-36-4, 31119-53-6)
12 alternate names
10 safety hazards
3 suppliers
POTASSIUM NITRITE
(7758-09-0)
12 alternate names
4 safety hazards
3 suppliers
NICKEL SULFATE
(7786-81-4, 10101-97-0, 10101-98-1, 7786-81-4, 10101-97-0)
12 alternate names
7 safety hazards
3 suppliers
bicarbonate
(71-52-3)
12 alternate names
3 suppliers
Iron(II) sulfate
(7720-78-7, 7782-63-0, 13463-43-9, 7720-78-7, 16547-58-3)
12 alternate names
2 safety hazards
3 suppliers
ZINC ACETATE
(557-34-6)
12 alternate names
3 suppliers
Lead(II) sulfate
(52732-72-6, 90583-07-6, 15739-80-7, 7446-14-2)
12 alternate names
5 safety hazards
3 suppliers
POTASSIUM PERMANGANATE
(7722-64-7)
12 alternate names
6 safety hazards
3 suppliers
ammonium hydroxide
(7664-41-7, 1336-21-6)
12 alternate names
3 safety hazards
3 suppliers
AMMONIUM BIFLUORIDE
(1341-49-7)
12 alternate names
1 safety hazard
3 suppliers
Ethide
(594-72-9)
12 alternate names
4 safety hazards
3 suppliers
Fluoroboric acid
(16872-11-0)
12 alternate names
3 safety hazards
3 suppliers
POTASSIUM ARSENATE
(7784-41-0)
12 alternate names
6 safety hazards
3 suppliers
CAS Directory